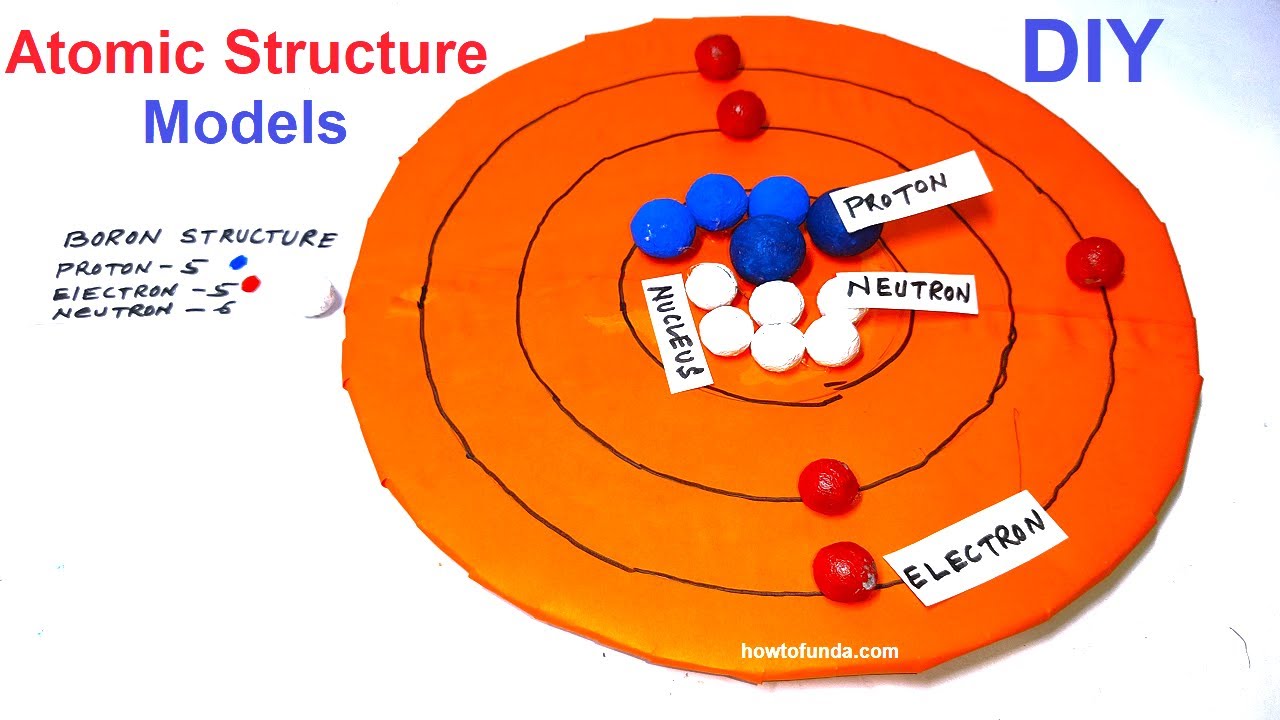
Understanding Bohr's model requires some knowledge of electromagnetic radiation (or light).īohr's key idea in his model of the atom is that electrons occupy definite orbitals that require the electron to have a specific amount of energy. This nucleus is surrounded by two-electron shells named K-shell and L-shell. In 1913, the Danish physicist Niels Bohr proposed a model of the electron cloud of an atom in which electrons orbit the nucleus and were able to produce atomic spectra. The Bohr Model of Boron (B) has a nucleus that contains 6 neutrons and 5 protons. These difficulties cast a shadow on the planetary model and indicated that, eventually, it would have to be replaced. 
The number of protons, as well as the number of electrons in an atom, is always equal to its atomic number. Furthermore, Rutherford's model was unable to describe how electrons give off light forming each element's unique atomic spectrum. The Bohr model of Beryllium consists of 4 protons and 5 neutrons in its nucleus, while 4 electrons revolve around the nucleus. If the electron circling the nucleus in an atom loses energy, it would necessarily have to move closer to the nucleus as it loses energy, and would eventually crash into the nucleus. This is, after all, how we produce TV signals. It does introduce several important features of all models used to describe the distribution of electrons in an atom. It was already known that when a charged particle (such as an electron) moves in a curved path, it gives off some form of light and loses energy in doing so. The Bohr model is a relatively primitive model of the hydrogen atom, compared to the valence shell model. Bohr’s model of the hydrogen atom provides insight into the behavior of matter at the microscopic level, but it does not account for electronelectron interactions in atoms with more than one electron. Unfortunately, there was a serious flaw in the planetary model. It was already known that when a charged particle (such as an electron) moves in a curved path, it gives off some form of light and loses energy in doing so. The fourth and fifth orbits would therefore hold 18 electrons. 8 elements in the third row, 8 electrons in the third orbit. 8 elements in the second row, 8 electrons in the second orbit. 2 elements in the first row, 2 electrons in the first orbit. \): Niels Bohr with Albert Einstein at Paul Ehrenfest's home in Leiden (December 1925). 3: Niels Bohr with Albert Einstein at Paul Ehrenfests home in Leiden (December 1925). The Bohr model arrangement of electrons follows the same pattern as the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
